Archived content
NOTE: this is an archived page and the content is likely to be out of date.
Fujitsu Launches SaaS Solution for Clinical Drug Trials
- Delivering on-demand, low cost IT resources according to trial length and scale -
Fujitsu Limited,Fujitsu System Solutions Limited
Tokyo, January 19, 2010
Fujitsu Limited and Fujitsu System Solutions Limited today announced the launch of software as a service (SaaS)-based solution to support the operation of clinical drug trials conducted by pharmaceutical companies. The new service, called tsClinical, is now available in Japan.
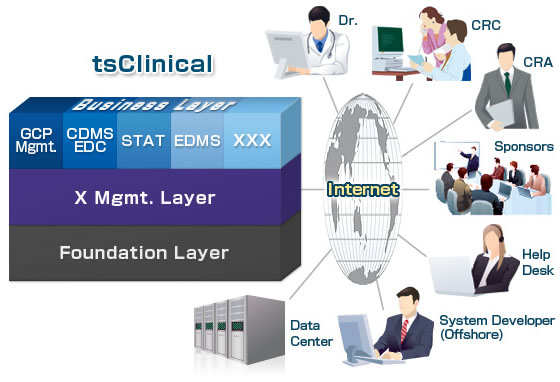
tsClinical is an on-demand SaaS-based solution that provides a data management system for results from medical cases necessary for clinical trials of new drugs, according to trial length and scale, through a network.
The new service eliminates the need for users to deploy an individual case data management system for each clinical trial, thus speeding up the trial process, eliminating system deployment and maintenance costs, and minimizing the workload associated with clinical trials.
tsClinical has already been deployed by Bellsystem24, Inc., a Japan-based organization which provides support services for clinical trials. In addition to existing support services, Bellsystem24 aims to deliver enhanced CRO(1) services by providing new services, connecting its pharmaceutical customers with a data management environment through SaaS.
Pharmaceutical companies, healthcare institutions, and CROs have been conducting a variety of clinical trials to test new drugs for lifestyle-related diseases, infectious diseases, and healthcare maintenance, as well as to determine the side effects of drugs already on the market. However, during these clinical trials, it is important for companies to collect and manage data containing information on the effectiveness or side effects of a drug, both quickly and accurately. Companies must also employ a case data management system that complies with related regulations for each respective clinical trial. As a result, it has become a challenge for pharmaceutical companies to quickly deploy case data management systems to conduct fast clinical trials while curtailing system development costs.
To optimize clinical trial operations during new drug development, Fujitsu is offering an on-demand SaaS-based data management system, which previously was only available as a software package.
Overview
tsClinical is composed of a three-layer structural framework which includes operations support (business layer), across-the-board management support (X management layer), and a data and services platform (foundation layer). As a first step, Fujitsu will be offering a package of data management services that combine both business and foundation layers, to gradually expand its offerings in the future.
- Tailored-use according to the duration of the clinical trial
Fujitsu delivers the SaaS version of a well-established software package, previously available for use only through the purchase of a license, allowing users to contract for the required services only for as long as they are needed. As a result, users can curb their spending on system and operation management for periods during which clinical trials are not being conducted.
- Minimizes the validation process required for documentation of facilities, procedures, and schedules essential for production and quality control
Fujitsu provides an environment that significantly reduces the validation process for documentation that, up until now, customers had to internally do on their own.
- Simultaneous use by multiple companies or organizations
As the SaaS-based solution allows the pharmaceutical company and its CRO to use the same IT environment when conducting a clinical trial, data sharing and progress management becomes easier thus enhancing collaborative efforts.
List of Services
| Service Menu | Overview |
|---|
| Data management | Basic service utilizing the case data management system |
| Protocol operations | Operations service fee for one clinical trial |
| Option for customized input | Delivers form output functionality |
| Option for case progress management | Delivers case progress management functionality |
| Option for adding additional accounts | Allows users to add additional accounts |
| Data archiving | Allows users to archive data in XML format |
Pricing and Availability
| Service Name | Pricing (excluding tax) | Availability in Japan |
|---|
| tsClinical | 1 test: starting from JPY 150,000 per month | Available now |
* Pricing will differ according to the number of services deployed.
Sales Target
JPY 1 billion over the next three years.
About Fujitsu
Fujitsu is a leading provider of IT-based business solutions for the global marketplace. With approximately 175,000 employees supporting customers in 70 countries, Fujitsu combines a worldwide corps of systems and services experts with highly reliable computing and communications products and advanced microelectronics to deliver added value to customers. Headquartered in Tokyo, Fujitsu Limited (TSE:6702) reported consolidated revenues of 4.6 trillion yen (US$47 billion) for the fiscal year ended March 31, 2009. For more information, please see: www.fujitsu.com.
Press Contacts
Public and Investor Relations Division
Inquiries
Company:Fujitsu Limited
All other company or product names mentioned herein are trademarks or registered trademarks of their respective owners. Information provided in this press release is accurate at time of publication and is subject to change without advance notice.
This press release has been revised as of December 17, 2018.
Date: 19 January, 2010
City: Tokyo
Company:
Fujitsu Limited,
Fujitsu System Solutions Limited,
,
,
,